How do you stack up against your peers with CMS requests?
Category: General
We were thrilled to see such a great response to our CMS Audits survey. We asked you to help us understand what volume of audits you face on a monthly basis. We received 125 responses with very interesting results that give insight into CMS Audits.
Over half of respondents (58% total) receive 5 or fewer requests for additional documentation each month, but 14% – that’s 1 in 7 respondents! – must coordinate responses to over 20 requests per month.
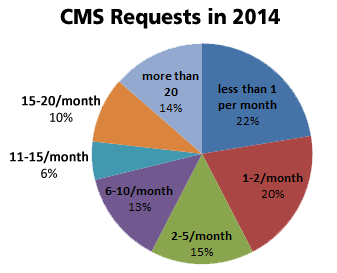
Our respondent demographics skewed toward DME/HME providers, with over 80% of the completed surveys coming from that area of the healthcare industry. DME/HME providers were well represented within each level of CMS request volume, with nearly even division across all categories.
The non-DME/HME providers we heard from came from a range of areas including Home Health, Pharmacy (LTC, specialty, and retail), Orthotics & Prosthetics, Sleep Lab, Nursing Home/Rehab and one Hospital. With the exception of a single outlier, Pharmacy providers received under five CMS requests per month in 2014. This was not entirely surprising as the process for medication reimbursement is heavy on prior authorization, which can reduce auditing concerns later on.
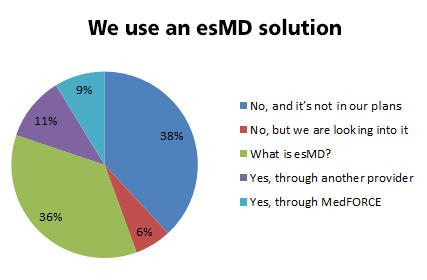
Only 12% of respondents currently use an esMD solution, while another 6% are actively pursuing engaging one. Over one third of respondents did not know what esMD was. This was the biggest surprise to us – and we urge all of you in this category to look into one, such as ZipMit, to dramatically ease the burden of CMS communications.
To learn more about ZipMit, MedFORCE's esMD solution, please request a demo today.

To learn more or request a demonstration of what Medforce software can do for you fill in this form or call our sales department at:
Even our demos let you preview without leaving your desk. View the power of Medforce right from the comfort of your office.